Currently not accepting new graduate students
- Member - Turkish Academy of Science
- Fellow - American Academy of Microbiology
- Fellow - American Association for the Advancement of Science
- DAAD Alumnus
- Ph.D., University of Strasbourg, France
- Post-Doc., Harvard Medical School
- Scientist, Cold Spring Harbor Laboratory
Photosynthetic and respiratory electron transfer pathways; Structure, function, and biogenesis of membrane cytochrome complexes
We are interested in understanding the molecular basis of biological electron transfer in photosynthesis and respiration. These basic metabolic pathways contain oligomeric, membrane-bound protein complexes with redox-active prosthetic groups (heme and copper atom). They are vital components for cellular functions ranging from ATP synthesis to protein secretion, solute transport, motility, and thermogenesis. Their dysfunction severely compromises cellular energy production and leads to low crop yields in plants (photosynthesis), or neurological and muscular diseases in humans (respiration). Complete understanding of how evolutionarily conserved energy producing molecular machines perform their functions is of considerable biological significance and general interest. Our studies aim to define the structure, function, assembly, biogenesis, and regulation of these proteins in response to environmental signals, including light and oxygen. Cytochrome bc1 complex and cytochrome cbb3 oxidase (membrane-associated proton pumps) and their physiological electron carriers (cytochromes c2 and cy) are subjects of our studies. The maturation of their cofactor (heme and copper) containing subunits, and assembly of mature components and their atomic structures are parts of our studies.
We use molecular genetics and omics combined with biochemical, biophysical, and structural (cryo-EM) approaches. We use the purple non-sulfur photosynthetic bacteria R. capsulatus and R. sphaeroides as model organisms instead of mitochondria of eukaryotes or chloroplasts of plants due to their experimental analyses. Current work is focused on the 1) Structure and function of the cytochrome super-complexes and assembly of their subunits; 2) Maturation (covalent addition of heme groups) of c-type cytochromes including cytochrome cy, cytochrome bc1, and cytochrome cbb3 oxidase; 3) Cellular copper trafficking and delivery to cuproenzymes.
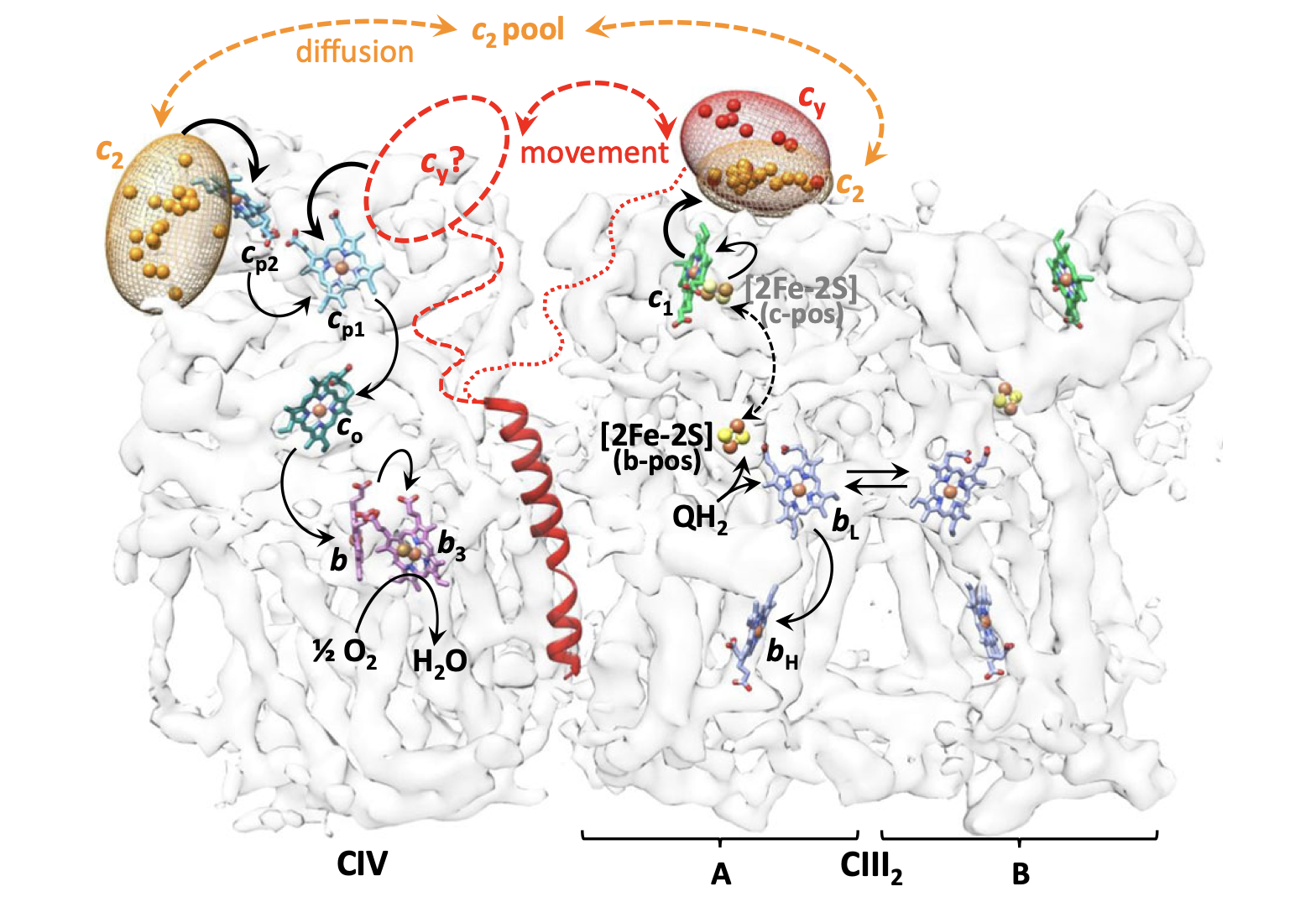
Cytochromes c2 and cy binding sites and electron transfer pathways around the bc1-cy-cbb3 super complex. The likely binding regions of c2 and cy (orange and red ellipsoids) are depicted by the distributions of their heme-Fe atoms on the bc1-cy-cbb3 super complex (transparent). The linker region of cy is indicated by dotted and dashed lines between its transmembrane and head domains. The electron pathways are shown by thick and thin black arrows. The double headed dashed black arrow indicates the movement of the FeS subunit of bc1. Note that cytochrome c2 is peripheral while cytochrome cy is integral to the membrane.
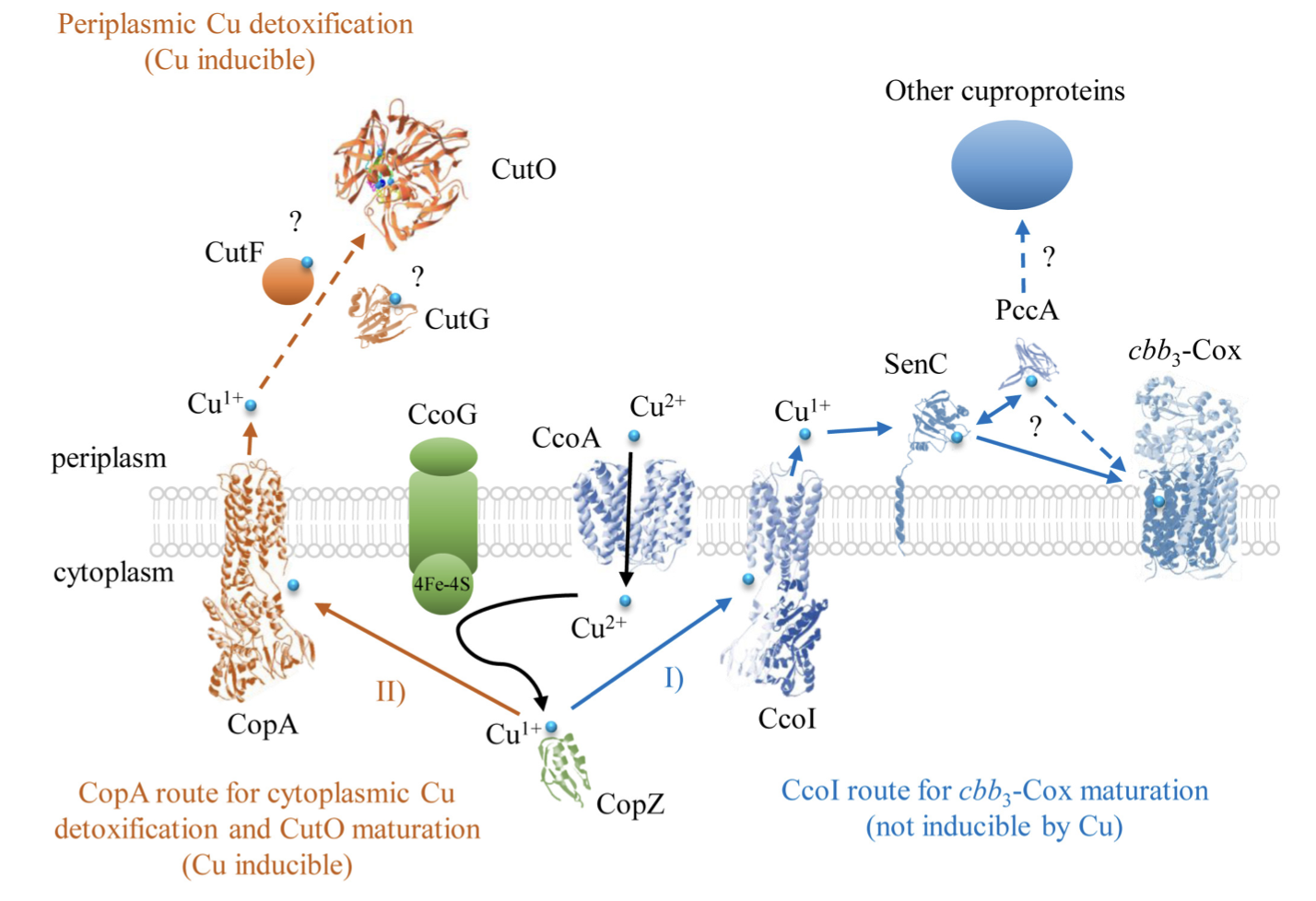
Copper (Cu) detoxification and cuproenzyme biogenesis pathways illustrated using R. capsulatus cytochrome cbb3-oxidase (cbb3-Cox) and multicopper oxidase (CutO). The two Cu export routes to the periplasm, one via CcoI and the other via CopA (both P1B-type ATPases) are shown. Cu bound to SenC/PccA is inserted to cbb3-Cox. Excess cytoplasmic Cu bound to CopZ is exported out. The periplasmic CutO partly receives its Cu via the CopZ-CopA pathway. PBD structures are 3RFU (CopA and CcoI), 6WIS (CopG for CutG), 3OD3 (CueO for CutO), 1K0V (CopZ), 3WDO (YajR for CcoA), 4WBR (ScoI/SenC), 2K70 (PCuAC/PccA) and 5DJQ (cbb3-Cox).
- BIOL 401 - Prokaryotic Microbiology
- BIOL 101 - Introductory Biology
Books:
Microbial Energy Transduction: Genetics, Structure and Function of Membrane Proteins. 1986. D. C. Youvan and F. Daldal, Eds. Cold Spring Harbor Laboratory Press, NY., USA.
The Purple Phototrophic Bacteria. 2008. C. N. Hunter, F. Daldal, M. C. Thurnauer and J. T. Beatty, Eds. Springer, Dordrecht, The Netherlands.
Selected Recent Publications (2014-2023):
Ekici, S., Turkarslan, S., Pawlik, G., Dancis, A., Baliga, N., Koch, H-G. and Daldal, F. (2014). Intracytoplasmic copper homeostasis controls cytochrome c oxidase production. MBio, 5: 1055-13. PMID: 2445735.
Verissimo, A. F. and Daldal, F. (2014) Cytochrome c maturation: an intricate process catalyzed by a supercomplex? Biochim. Biophys Acta Bioenergetics, 1837: 989-998. PMID: 24631867.
Yildiz, G. G., Gennis, R. B., Daldal, F. and Ozturk, M. (2014) The KC-channel in the cbb3-type respiratory oxygen reductase from Rhodobacter capsulatus is required for both chemical and pumped protons. J. Bacteriol. 196: 1825-1832. PMID: 24563037.
Verissimo, A. F., Shroff, N. and Daldal, F. (2015). During Cytochrome c Maturation CcmI Chaperones the Class I Apocytochromes until the Formation of Their b-Type Cytochrome Intermediates. J. Biol. Chem. 290: 16989-7003 . PMID: 25979338
Trasnea, P-I., Utz M., Khalfaoui-Hassani, B., Lagies, S., Daldal, F. and Koch, H-G. (2015). Cooperation between two periplasmic copper chaperones is required for full activity of the cbb3-type cytochrome c oxidase and copper homeostais in Rhodobacter capsulatus. Mol. Micro. 100: 345-61. PMID: 26718481.
Khalfaoui-HAssani, B., Verissimo, A. F., Shroff, N., Ekici, S., Trasnea, P-I, Utz, M., Koch, H-G, and Daldal, F. (2016) Biogensesis of cytochrome c complexes: from insertion of redox cofactors to assembly of different subunits, pp. 527-555. In Cytochrome Complexes: Evolution, Structures, Energy Transduction, and Signaling. Cramer, W. and Kallas, T. (Editors), Springer Dordrecht
Khalfaoui-Hassani, B., Verissimo, A. F., Koch, H-G, and Daldal, F. (2016). Uncovering the Transmembrane Metal Binding Site of the Novel Bacterial MFS-type Copper Importer CcoA. MBio. 7: 1981-15. PMID: 26787831
Francia, F., Malferrari, M., Lanciano, P., Daldal, F. and Venturoli, G. (2016). The Cytochrome b Zn binding Amino Acid Residue Histidine 291 is Essential for Ubihydroquinone Oxidation at the Qo Sute of Bacterial Cytochrome bc1. Biochim. Biophys Acta-Bionergetics 1857: 1796-1806. PMID: 27550309
Vos, M. H., Reeder, B. J., Daldal, F. and Liebl, U. (2017). Ultrafast photochemistry of the bc1 complex. Phy. Chem. Chem. Phys. 19: 6807-6813. PMID: 28218331 and correction.
Verissimo AF, Khalfaoui-Hassani B, Hwang J, Steimle S, Selamoglu N, Sanders C, Khatchikian CE, Daldal F. (2017). The thireduction component CcmG confers efficiency and the hem ligation component CcmH ensures stereo-specificty during cytochrome c maturation. J. Biol. Chem. 292: 13154-13167. PMID: 28634234
Onder, O., Verissimo, AF., Khalfaoui-Hassani, B., Peters, A., Koch, H-G. and Daldal, F. (2017). Absence of thiol-disulfide oxidoreductase DsbA impairs cbb3-type cytochrome c oxidase biogenesis in Rhodobacter capsulatus. Fron. Microbial. 8: 2576. PMID:29312253
Tropeano, CV., Fiori, J., Carelli, V. Caporali, L., Daldal, F., Ghelli, AM and Rugolo, M. (2018). Complex II phosphrylation is triggered by unbalanced redox homeostasis in cells lacking Complex III. Biochim Biophys Acta Bioenergetics, 1859: 182-190. PMID: 29269267 and corrigendum.
Khalfaoui-Hassani, B., Wu, H., Blaby-Haas, C., Zang, Y., Sandir, F., Verissimo, AF., Koch, H-G. and Daldal, F. (2018). Widespread distribution and functional specificity of the copper imprter CcoA: Separate copper uptake and delivery routes to bacterial cytochrome c oxidases. MBio, 9: e00065-18. PMID: 29487231
Sandri, F., Musiani, F., Selamogly, N., Daldal, F. and Zannoni, D. (2018). Pseudomonas pseudoalcaligenes KF707 grown with biphenyl expresses a caa3 type cytochrome c oxidase. FEBS Lett 1873: 3468- PMID: 29427514
Trasnea, P-I., Andreei, A., Marckmann, D, Utz, M., Khalfaoui-HAssani, B., Daldal, F. and Koch, H-G. (2018). Copper tranfer between two periplasmic copper chaperones involved in Rhodobacter capsulatus cbb3-type cytochrome oxidase biogenesis. ACS Chem. Biol. 13: 1388-1397. PMID: 29613755
Utz, M., Andreo, A., Milanov, M., Trasnea, P-I., Marckmann, D., Daldal, F. and Koch, H-G. (2019). The Cu chaperone CopZ is required for Cu homeostasis in Rhodobacter capsulatus and influences cytochrome cbb3 oxidase assembly. Mol. Micro. 111: 764-783.
Zang, Y., Blaby-Haas, C. E., Steimle, S., Verissimo, A. F., Garcia-Angulo, V. A., Koch, H-G., Daldal, F. (corresp. auth), and Khalfaoui-Hassani, B. (2019). Cu Transport by the Extended Family of CcoA-like Transporters (CalT) in Proteobacteria. Science Report, 9: 1208.
Marckmann, D., Trasnea, P-I., Schimpf, J., Winterstein, C., Andrei, A., Blaby-Haas, C. E., Friedrich, T., Daldal, F. (corres. auth), and Koch, H-G. (2019). The cbb3-type cytochrome oxidase assembly factor CcoG is a widely distributed novel cupric reductase. Proc. Nat. Acad. Sci., 116: 21166-21175.
Tropeano, C.V., Aleo, S.J., Zanna, C., Roberti, M., Scandiffio, L., Cantatore, P.,Loguercio Polosa, P., Fiori, J., Porru, E., Roda, A., Carelli, V., Steimle, S., Daldal, F., Rugolo, M. and Ghelli, A. (2020). Fine-tuning of the respiratory complexes stability and supercomplexes assembly in cells defective of complex III. Biochim Biophys Acta Bioenergetics 1861:148133.
Selamoglu, N., Önder, O., Ozturk, Y., Khalfaoui-Hassani, B., Garcia, B., Koch, H-G. and Daldal, F. (2020) Comparative Differential Cuproproteomes of Rhodobacter capsulatus Reveal Novel Copper Homeostasis Related Proteins. Metallomics, 12: 572-591.
Andrei, A., Öztürk, Y., Khalfaoui-Hassani, B., Rauch, J., Marckmann, D., Trasnea, P-I., Daldal, F., and Koch, H-G. (2020). Cu Homeostasis in Bacteria: The Ins and Outs. Membranes 10: 242-287.
Steimle, S., VanEeuwen, T., Ozturk, Y., Kim, H-J., Braitbard, M., Garcia, B.A., Schneidman-Duhovny, D., Murakami, K. (corres. auth) and Daldal, F. (corres. auth). (2021). Cryo-EM Structure of Respiratory bc1-cbb3 type CIII2CIV Supercomplex and Electronic Communication Between its Complexes. Nature Communications, 12: 929-944.
Khalfaoui-Hassani, B., Trasnea, P-I., Steimle, S. Koch, H-G., and Daldal. F. Cysteine Mutants of the Major Facilitator Superfamily-Type Transporter CcoA Provide Insight into Cu Import. (2021). MBio, 12: e01567-21.
Andreea Andrei, A., Di Renzo, M.A, Öztürk, Y., Meisner, A., Daum, N., Frank, F., Rauch, J., Daldal, F., Andrade, S.L.A and Koch, H-G. (2021). The CopA2-type P1B –type ATPase CcoI serves as central hub for cbb3-Cytochrome Oxidase biogenesis. (2021). Fron. Microbiol. 12: 712465.
Öztürk, Y., Blaby-Haas, C.E., Daum, N., Andrei, A., Rauch, J., Daldal, F. (corres. auth). and Koch, H-G. (corres. auth). (2021). Maturation of Rhodobacter capsulatus Multicopper Oxidase CutO Depends on the CopA Copper Efflux Pathway and Requires the cutF Product. Front. Microbiol. 12: 720644.
Paquette, C. M., Nitschke, W., Daldal, F. and Zannoni, D. (2021). Editorial: Microbial Bioenergetics. Front Microbiol. 12: 793917.
Öztürk, Y., Andrei, A., Blaby-Haas, C.E., Daum, N., Daldal, F. (corres. auth) and Koch, H-G. (corres. auth). (2023). Metabolic Sensing of Extracytoplasmic Copper Availability via Translational Control by a Nascent Exported Protein. MBio, 14: 3040-22.
Cell and Molecular Biology Graduate Group - https://www.med.upenn.edu/camb/index.shtml
Biochemistry and Molecular Biophysics Graduate Group - http://bio-sharp-001.med.upenn.edu

